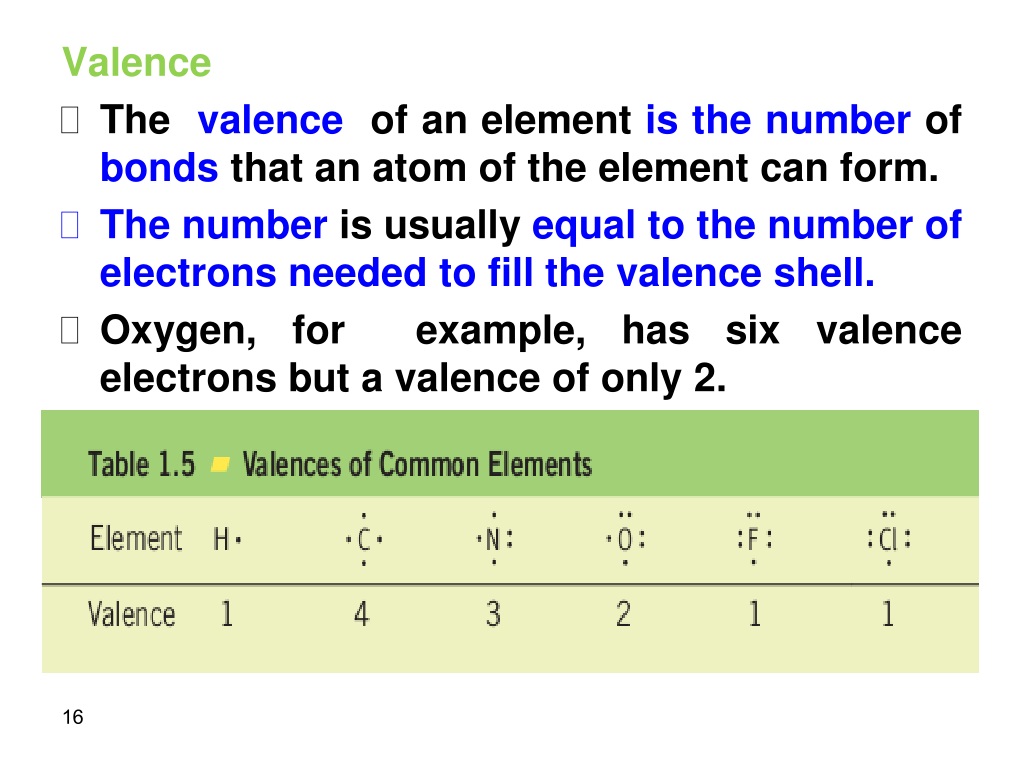
The problem with this structure is that boron has an incomplete octet it only has six electrons around it. If one was to make a Lewis structure for BH 3 following the basic strategies for drawing Lewis structures, one would probably come up with this structure (Figure 3.8.3): Figure 3.8.3 Let's take a look at one such hydride, BH 3 (Borane). Species with incomplete octets are pretty rare and generally are only found in some beryllium, aluminum, and boron compounds including the boron hydrides. This is also the case with incomplete octets. There are even more occasions where the octet rule does not give the most correct depiction of a molecule or ion. The second exception to the Octet Rule is when there are too few valence electrons that results in an incomplete Octet. That is exactly what is done to get the correct Lewis structure for nitrogen monoxide (Figure 3.8.2): Figure 3.8.2: The proper Lewis structure for NO molecule However, if we add the eleventh electron to nitrogen (because we want the molecule to have the lowest total formal charge), it will bring both the nitrogen and the molecule's overall charges to zero, the most ideal formal charge situation. The overall molecule here has a formal charge of +1 (+1 for nitrogen, 0 for oxygen. Oxygen therefore has a formal charge of 0. In Figure 3.8.1, oxygen has four lone pair electrons and it participates in two bonds with nitrogen. Oxygen normally has six valence electrons. This results in nitrogen having a formal charge of +1. In Figure 3.8.1, it has two lone pair electrons and it participates in two bonds (a double bond) with oxygen. Nitrogen normally has five valence electrons. Let's look at the formal charges of Figure 3.8.2 based on this Lewis structure. If we were to imagine nitrogen monoxide had ten valence electrons we would come up with the Lewis Structure (Figure 3.8.1): Figure 3.8.1: This is if nitrogen monoxide has only ten valence electrons, which it does not. If you need more information about formal charges, see Lewis Structures. Nitrogen monoxide has 11 valence electrons. An example of a stable molecule with an odd number of valence electrons would be nitrogen monoxide. No formal charge at all is the most ideal situation. The formal charge is the perceived charge on an individual atom in a molecule when atoms do not contribute equal numbers of electrons to the bonds they participate in. But where should the unpaired electron go? The unpaired electron is usually placed in the Lewis Dot Structure so that each element in the structure will have the lowest formal charge possible. The lone electron is called an unpaired electron. The Octet Rule for this molecule is fulfilled in the above example, however that is with 10 valence electrons. The total would be 11 valence electrons to be used. 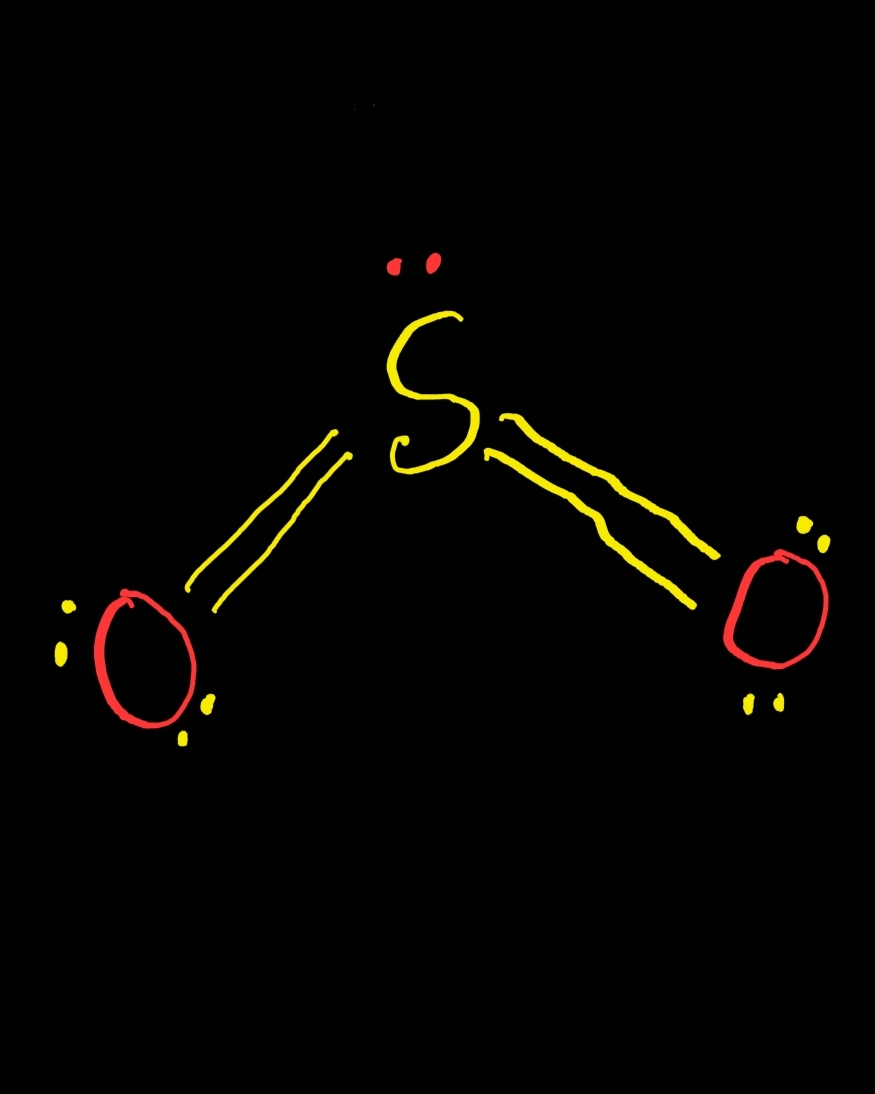
Nitrogen has 5 valence electrons while Oxygen has 6. An example of this would be Nitrogen (II) Oxide (NO ,refer to figure one). The first exception to the Octet Rule is when there are an odd number of valence electrons. 
\)Įxception 1: Species with Odd Numbers of Electrons
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
